The honey bee mating system is not just respectfully elaborate, it’s unbelievably dramatic.
I spend most of my day thinking about sperm. Ah, the life of a scientist: The profession authorizes us to deeply muse topics which would otherwise be quirky, fringe, or inappropriate. Scientists are just highly educated weirdos, really. And this freedom lets us unearth new chapters to stories we thought we knew by heart. I thought I understood how honey bee sex worked, but I was very, very wrong.
Just over a year ago, I started a postdoctoral fellowship with Dr. Jeff Pettis and Dr. David Tarpy, where I switched from studying social immunity to queen quality and reproductive health. Going into it, I knew that queens mated with around twelve to fifteen different drones, on average. I knew that queens stored millions of sperm in their spermathecae for the rest of their lives. And I knew that mating is a suicidal act for the poor drones, whose reputation is often diminished to nothing more than flying sperm vectors. What I didn’t know is that although the drone’s life may end when he mates, his influence is far from over.
Somewhat deviously, the drone’s seminal fluid manipulates queens to halt their promiscuous mating habits by making them go partially blind. Molecules in the semen somehow control important genes for eyesight in the queen’s brain, so they can’t see well enough to engage in more mating flights. Meanwhile, the queen’s reproductive tract is a war zone, with the sperm from different drones engaging in molecular warfare to incapacitate each other before they get stored. Bee sex is so much more dramatic than I ever imagined.
Now you see me, now you don’t
Dr. Joanito Liberti, a postdoctoral fellow at the University of Lausanne, Switzerland, and his coauthors discovered this unusual relationship between queens receiving semen and losing some parts of their eyesight. But they weren’t the first researchers to be curious about this topic. Others have found that mating induces dramatic structural rearrangements in the queen’s brain, which are accompanied by changes in neurotransmitters and gene expression.1-3 Liberti and his colleagues, though, are the first to show that receiving seminal fluid alone is sufficient to initiate changes in the brain, and that eyesight is one of the major biological processes that’s targeted.
In a paper recently published in “eLife,”4 Liberti describes how they used a technique called “electroretinography” to measure the nerve activity, induced by flashing light, in queens’ compound eyes and ocelli (three simple light-receptors on the forehead) after inseminating them with seminal fluid, semen, or saline. They found that queens receiving seminal fluid or semen had lower retinal nerve activity and were worse at finding their way home than queens receiving saline (if they continued to engage in subsequent mating flights). Presumably, their impaired vision caused them to get lost along the way, or possibly fail to find the drone congregation areas in the first place. Using a genetic technique called RNA-seq, they showed that receiving seminal fluid was sufficient to alter expression of genes related to photoreception in the queen’s brain.
Liberti says that discovering that vision-related genes were specifically affected by seminal fluid is the most fascinating aspect of the study. “It imposes a cost on queens during their subsequent mating flights by altering their visual perception,” he says. While additional mating flights increase the cumulative genetic diversity of sperm the queen stores, those flights are also dangerous. Risks from predators like birds or dragonflies, a turn in weather, or the added energy expenditure counterbalance the benefits of increased diversity. “The average number of flights that queens typically embark on is a balanced compromise between these costs and benefits,” Liberti says. “By increasing the total costs of flying via their manipulation of queen visual perception, males are shifting the balance towards even fewer flights for the queen.”
The idea that insemination could impair a queen’s eyesight sounds a little far-fetched, and Liberti admits that exactly how the seminal fluid achieves this is unclear. But this finding begins to explain the mechanism behind a phenomenon that many of us are familiar with: that virgin queens are phototactic (attracted to light), and mated queens prefer the dark. But how could this improbable phenomenon have evolved?
The more mating flights (and mates) a queen has, the smaller an individual drone’s genetic contribution to his future colony, and the lower the chance that his genes will end up in subsequent queens. Dawkins’ “The Selfish Gene” taught us that natural selection favours outcomes that propagate an individual’s genetic material, so it makes sense that drones have evolved selfish mechanisms of manipulating queens to have fewer mates than they might otherwise prefer. Early on in their evolution, drones that happened to have the right seminal fluid recipe to manipulate queens to have fewer mates would, as a consequence, contribute proportionally more genes to their ensuing colonies, and be more likely to pass those genes on to future queens. These queens in turn pass them on to future drones, and so, the frequency of this phenomenon rises in the population over time to become widespread as it is today.
But if having fewer rivals gives drones a better chance of passing on their genes, why don’t honey bees regress back to the monogamous methods of their ancestors? Because genetic diversity is a very good thing. Genetically diverse colonies are more successful, more adaptable, and more resilient. And after all, aside from emergencies and supersedures, a drone can only pass on his genes if the colony is successful enough to produce drones or swarm (or both). Despite being selfish, it’s actually in his best interest to allow some rival mates to persist. But queen manipulation isn’t the only way that drones can compete for genetic real estate after death.
Molecular wars
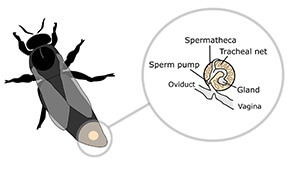
The process of sperm storage takes days after mating, and in the meantime, sperm from several rival mating partners coexists in the queen’s reproductive tract (first in the oviducts, then moving to the spermatheca). But it is not a benevolent coexistence: Seminal fluid actually contains molecular weapons that selectively incapacitate rival sperm.
Dr. Susanne den Boer, from the University of Copenhagen, Denmark, showed that adding secretions from accessory glands (which produce the proteins for seminal fluid) to a sperm solution killed the sperm if the secretion was from a rival male, but not when the secretion came from the male’s own glands.5 Furthermore, this was only true for….


